rongsheng jin for sale

Jahid S, Ortega JA, Vuong LM, Acquistapace IM, Hachey SJ, Flesher JL, La Serra MA, Brindani N, La Sala G, Manigrasso J, Arencibia JM, Bertozzi SM, Summa M, Bertorelli R, Armirotti A, Jin R, Liu Z, Chen CF, Edwards R, Hughes CCW, De Vivo M, Ganesan AK. PMID: 35385746; PMCID: PMC9127750.
Chen P, Zeng J, Liu Z, Thaker H, Wang S, Tian S, Zhang J, Tao L, Gutierrez CB, Xing L, Gerhard R, Huang L, Dong M, Jin R. PMID: 34145250; PMCID: PMC8213806.
Chen P, Lam KH, Liu Z, Mindlin FA, Chen B, Gutierrez CB, Huang L, Zhang Y, Hamza T, Feng H, Matsui T, Bowen ME, Perry K, Jin R. PMID: 31308519; PMCID: PMC6684407.
Lam, K. H., Guo, Z., Krez, N., Matsui, T., Perry, K., Weisemann, J., Rummel, A., Bowen, M. E. & Jin, R. A viral-fusion-peptide-like molecular switch drives membrane insertion of botulinum neurotoxin A1. Nat Commun 9, 5367 (2018) doi: 10.1038/s41467-018-07789-4.
Chen, P., Tao, L., Liu, Z., Dong, M. & Jin, R. Structural insight into Wnt signaling inhibition by Clostridium difficile toxin B. FEBS J (2018) doi: 10.1111/febs.14681.
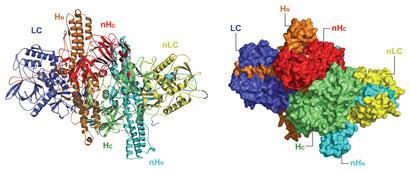
Chen, P., Tao, L., Wang, T., Zhang, J., He, A., Lam, K. H., Liu, Z., He, X., Perry, K., Dong, M*. & Jin, R*. Structural basis for recognition of frizzled proteins by Clostridium difficile toxin B. Science 360, 664-669 (2018) (*corresponding authors) doi: 10.1126/science.aar1999. PMCID: PMC6231499
Lam, K. H., Sikorra, S., Weisemann, J., Maatsch, H., Perry, K., Rummel, A., Binz, T. & Jin, R. Structural and biochemical characterization of the protease domain of the mosaic botulinum neurotoxin type HA. Pathog Dis 76 (2018) doi: 10.1093/femspd/fty044. PMCID: PMC5961070
Silva, D. A., Stewart, L., Lam, K. H., Jin, R. & Baker, D. Structures and disulfide cross-linking of de novo designed therapeutic mini-proteins. FEBS J 285, 1783-1785 (2018) doi: 10.1111/febs.14394. PMCID: PMC6001749
Lam, K. H., Qi, R., Liu, S., Kroh, A., Yao, G., Perry, K., Rummel, A. & Jin, R. The hypothetical protein P47 of Clostridium botulinum E1 strain Beluga has a structural topology similar to bactericidal/permeability-increasing protein. Toxicon 147, 19-26 (2018) doi: 10.1016/j.toxicon.2017.10.012. PMCID: PMC5902665
Chevalier, A., Silva, D.A., Rocklin, G.J., Hicks, D.R., Vergara, R., Murapa, P., Bernard, S.M., Zhang, L., Lam, K.H., Yao, G., Bahl, C.D., Miyashita, S.I., Goreshnik, I., Fuller, J.T., Koday, M.T., Jenkins, C.M., Colvin, T., Carter, L., Bohn, A., Bryan, C.M., Fernández-Velasco, D.A., Stewart, L., Dong, M., Huang, X., Jin, R., Wilson, I.A., Fuller, D.H. & Baker, D. Massively parallel de novo protein design for targeted therapeutics. Nature 550(7674):74-79 (2017) doi: 10.1038/nature23912. PMCID: PMC5802399
Yao, G., Lam, K.H., Weisemann, J., Peng, L., Krez, N., Perry, K., Shoemaker, C.B., Dong, M., Rummel, A. & Jin, R. A camelid single-domain antibody neutralizes botulinum neurotoxin A by blocking host receptor binding. Sci Rep. 7;7(1):7438. (2017) doi: 10.1038/s41598-017-07457-5. PMCID: PMC5547058
Yao, G., Lam, K.H., Perry, K., Weisemann, J., Rummel, A. & Jin, R. Crystal Structure of the Receptor-Binding Domain of Botulinum Neurotoxin Type HA, Also Known as Type FA or H. Toxins (Basel) 9, 93 (2017) doi: 10.3390/toxins9030093. PMCID: PMC5371848
Yao, G., Zhang, S., Mahrhold, S., Lam, K. H., Stern, D., Bagramyan, K., Perry, K., Kalkum, M., Rummel, A.*, Dong, M.* & Jin, R.* N-linked glycosylation of SV2 is required for binding and uptake of botulinum neurotoxin A. Nat Struct Mol Biol 23 (7):656-662 (2016) (*corresponding authors) doi: 10.1038/nsmb.3245. PMCID: PMC5033645
Lee, K., Lam, K. H., Kruel, A. M., Mahrhold, S., Perry, K., Cheng, L. W., Rummel, A. & Jin, R. Inhibiting oral intoxication of botulinum neurotoxin A complex by carbohydrate receptor mimics. Toxicon 107, 43-49 (2015) doi: 10.1016/j.toxicon.2015.08.003. PMCID: PMC4658216
Lam, K.H. & Jin, R. Architecture of the botulinum neurotoxin complex: a molecular machine for protection and delivery. Current Opinion in Structural Biology 31:89-95 (2015) doi: 10.1016/j.sbi.2015.03.013. PMCID: PMC4476938
Lam, K.H., Yao, G. & Jin, R. Diverse binding modes, same goal: The receptor recognition mechanism of botulinum neurotoxin. Progress in Biophysics and Molecular Biology 117(2-3):225-31 (2015) doi: 10.1016/j.pbiomolbio.2015.02.004. PMCID: PMC4417461
Lam, T.I., Stanker, L.H., Lee, K., Jin, R. & Cheng, L.W. Translocation of botulinum neurotoxin serotype A and associated proteins across the intestinal epithelia. Cellular Microbiology 17(8):1133-1143 (2015) doi: 10.1111/cmi.12424. PMCID: PMC4610714
Matsui, T.*, Gu, S., Lam, K.H., Carter, L.G., Rummel, A., Mathews, II. & Jin, R.* Structural Basis of the pH-Dependent Assembly of a Botulinum Neurotoxin Complex. J. Mol. Biol. 426(22):3773-3782 (2014) doi: 10.1016/j.jmb.2014.09.009. (*corresponding authors) PMCID: PMC4252799
Lee, K., Zhong, X., Gu, S., Kruel, A.M., Dorner, M.B., Perry, K., Rummel, A., Dong, M. & Jin, R. Molecular basis for disruption of E-cadherin adhesion by botulinum neurotoxin A complex. Science 344(6190):1405-1410 (2014) doi: 10.1126/science.1253823. PMCID: PMC4164303
Lee, K., Lam, K.H., Kruel, A.M., Perry, K., Rummel, A. and Jin, R. High-resolution crystal structure of HA33 of botulinum neurotoxin type B progenitor toxin complex. Biochem. Biophys. Res. Commun. 446(2):568-573 (2014) doi: 10.1016/j.bbrc.2014.03.008. PMCID: PMC4020412
Yao, Y., Lee, K., Gu, S., Lam, K.H. & Jin, R. Botulinum Neurotoxin A Complex Recognizes Host Carbohydrates through Its Hemagglutinin Component, Toxins (Basel) 6(2):624-635 (2014) doi: 10.3390/toxins6020624. PMCID: PMC3942755
Lee, K., Gu, S., Jin, L., Le, T.T.N., Cheng, L.W., Strotmeier, J., Kruel, A.M., Yao, G., Perry, K., Rummel, A.* & Jin, R.* Structure of a Bimodular Botulinum Neurotoxin Complex Provides Insights into Its Oral Toxicity. PLoS Pathog. 9(10): e1003690 (2013) doi:10.1371/journal.ppat.1003690. (*corresponding authors) PMCID: PMC3795040
Zong, Y. and Jin, R. Structural mechanisms of the agrin-LRP4-MuSK signaling pathway in neuromuscular junction differentiation. Cell. Mol. Life Sci. 70(17):3077-88 (2013) doi: 10.1007/s00018-012-1209-9. PMCID: PMC4627850
Gu, S. and Jin, R. Assembly and function of the botulinum neurotoxin progenitor complex. Curr. Top. Microbiol. Immunol. 364:21-44 (2013) doi: 10.1007/978-3-642-33570-9_2. PMCID: PMC3875173
Gu, S., Rumpel, S., Zhou, J., Strotmeier, J., Bigalke, H., Perry, K., Shoemaker, C.B., Rummel, A. & Jin, R. Botulinum neurotoxin is shielded by NTNHA in an interlocked complex. Science 335(6071):977-81 (2012) doi: 10.1126/science.1214270. PMCID: PMC3545708
Zong, Y., Zhang, B., Gu, S., Lee, K., Zhou, J., Yao, G., Figueiredo, D., Perry, K., Mei, L.* & Jin, R.* Structural basis of neuron-specific regulation of postsynaptic differentiation. Gene & Development 26:247-258 (2012) doi: 10.1101/gad.180885.111. (*corresponding authors) PMCID: PMC3278892
Yao, G., Zong, Y., Gu, S., Zhou, J., Xu, H., Mathews, II. & Jin, R. Crystal structure of the glutamate receptor GluA1 amino-terminal domain. Biochem. J. 438(2):255-63 (2011) doi: 10.1042/BJ20110801. PMCID: PMC3296483
Strotmeier, J., Gu, S., Jutzi, S., Mahrhold, S., Zhou, J., Pich, A., Eichner, T., Bigalke, H., Rummel, A.*, Jin, R.* & Binz, T*. The biological activity of botulinum neurotoxin type C is dependent upon novel types of ganglioside binding sites. Mol. Microbiol. 81(1):143-56 (2011) doi: 10.1111/j.1365-2958.2011.07682.x. Epub 2011 Jun 2. (*corresponding authors)
Strotmeier, J., Lee, K., Völker, A.K., Mahrhold, S., Zong, Y., Zeiser, J., Zhou, J., Pich, A., Bigalke, H., Binz, T., Rummel, A.* & Jin, R.* Botulinum neurotoxin serotype D attacks neurons via two carbohydrate-binding sites in a ganglioside-dependent manner. Biochem. J. 431(2):207-16 (2010) (*corresponding authors)
Jin, R.*, Singh, S.K., Gu, S., Furukawa, H., Sobolevsky, A.I., Zhou, J., Jin, Y. & Gouaux E.* Crystal structure and association behavior of the GluR2 amino-terminal domain. EMBO J. 28(12):1812-23 (2009) (*corresponding authors) PMCID: PMC2699365
Kumar, J., Schuck. P., Jin, R. & Mayer, M.L. The N-terminal domain of GluR6-subtype glutamate receptor ion channels. Nat. Struct. Mol. Biol. 16(6):631-8 (2009) PMCID: PMC2729365
Jin, R., Rummel, A., Binz, T. & Brunger, A.T. Botulinum neurotoxin B recognizes its protein receptor with high affinity and specificity. Nature 444:1092-5 (2006)
Jin, R., Clark, S., Weeks, A.M., Dudman, J.T., Gouaux, E. & Partin, K.M. Mechanism of positive allosteric modulators acting on AMPA receptors. J. Neurosci. 25(39):9027-36 (2005)
Jin, R., Junutula, J.R., Matern, H.T., Ervin, K.E., Scheller, R.H. & Brunger, A.T. Exo84 and Sec5 are competitive regulatory Sec6/8 effectors to the RalA GTPase. EMBO J. 24:2064-74 (2005)
Jin, R., Bank, T., Mayer, M. L., Traynelis, S. & Gouaux, E. Structural basis for partial agonist action at ionotropic glutamate receptors. Nat. Neurosci. 6(8):803-10 (2003)

Rongsheng is an associate degree holder. Protective Service Occupations is listed as a current occupation. Rongsheng’s residency is at 2005 California Str, Mountain View, CA 94040. These are the name of three companies which are registered at this address: Xl Media Group and Wornit, LLC. Nick Hartmann, Michelle Solone, and 116 other persons are also associated with this address. (650) 962-9375 (Pacific Bell), (650) 224-3428 (New Cingular Wireless PCS, LLCPacific Bell) are the phone numbers associated with Rongsheng. The phone number (650) 962-9375 is also used by Argelia Recinos, Argelia Recino

Botulism is caused when the botulinum neurotoxin (BoNT) inhibits the release of a neurotransmitter. The disease can be caused by eating toxin-contaminated food, but how the BoNT protein survives the digestive tract and reaches the bloodstream has been a mystery. Last year, a group led by Rongsheng Jin of the University of California, Irvine, demonstrated how a protein called nontoxic nonhemagglutinin (NTNHA) binds to and shields BoNT to protect it from digestive proteases. Jin and colleagues have now used electron microscopy and X-ray crystallography to study a complex of BoNT, NTNHA, and three hemagglutinin proteins that play a role in getting BoNT past intestinal cells to the blood (PLoS Pathog. 2013, DOI: 10.1371/journal.ppat.1003690). The researchers find that the 760-kilodalton complex evokes the construction of the Apollo lunar lander, with BoNT and NTNHA on top and the hemagglutinins forming “legs,” which are the parts that interact with intestinal epithelial cells. The legs land on and bind to sugars on the cells, facilitating passage of BoNT. Jin and colleagues find that dosing mice with a monosaccharide can reduce BoNT toxicity, suggesting a way to prevent—but not treat—botulism. The full protein complex could also point to ways to deliver protein drugs orally.

Now, a team from the Sanford-Burnham Medical Research Institute in California and the Medical School of Hannover in Germany, led by Rongsheng Jin, has cracked the problem and identified how a ’bodyguard’ protein protects its BONT master before releasing it into bloodstream.
’We were surprised to see that NTNHA, which is not toxic, turned out to be remarkably similar to botulinum neurotoxin. It’s composed of three parts, just like a copy of the toxin itself. The two proteins hug each other and interlock with what looks like a handshake,’ says Jin. This tight grip serves to hide the sensitive portions of the toxin and protect them from attack by acid and by digestive enzymes.
The team also identified residues on the NTNHA protein that are sensitive to pH - effectively acting as pH sensors. ’In the low pH of the digestive tract, the bodyguard protein remains tightly interlocked with the toxin protein,’ says Jin. ’But when it enters the bloodstream and the pH rises, the structure changes and the toxin is released.’
Knowledge of how the bodyguard exerts its protective influence might allow small molecules to be developed that can interfere with its action and act as an antidote to the poison, Jin suggests. ’This is something we are looking into.’
(Bloomberg) — China Rongsheng Heavy Industries Group Holdings Ltd., once the country’s largest private shipyard, said it will not proceed with a proposed warrant sale after a potential investor who pledged as much as HK$3.2 billion ($413 million) was detained.
Wang’s detention occurred days before China Rongsheng shareholders are to meet March 13 to vote on the investment by Kingwin Victory. The firm agreed in October to pay at least HK$510 million for warrants Rongsheng is selling, and its total investment could reach HK$3.2 billion.
Rongsheng “has no information as to the details of the incident and has been unable to contact Mr. Wang Ping, which casts doubt” on Kingwin Victory’s ability to follow through on the investment by the March 31 deadline, Rongsheng said Wednesday in a statement to the Hong Kong Exchange.
The Beijing Public Security Bureau didn’t immediately respond to a faxed request for comment, and Wang couldn’t immediately be reached. Shares of the company fell as much as 8.1 percent in Hong Kong trade to HK$0.68 on Thursday, down from more than HK$8 in 2011. It was down 5.4 percent as of 9.35 a.m. local time.
Rongsheng has run into financial trouble amid competition with state-owned yards that have government backing and easier access to financing. The company is also seeking investment in its Jiangsu shipyard and bought a stake in an oilfield in Kyrgyzstan as part of efforts to restructure its business.
Shanghai-based Rongsheng said last August it’s entering the energy business and is issuing new shares so it can buy a 60 percent stake in a Kyrgyzstan oilfield. It later said it’s seeking to identify new investment opportunities outside China, including in Central Asia.
Botulinum toxin (Botox) is a large protein that is composed of three domains (upper right corner, crystal structure shown in yellow, green, and pink). The new study reveals a novel mechanism by which the toxin hijacks three receptors on the host neuronal surface as its “GPS” — the peptide moiety of protein SV2 (green-blue ribbon model), a conserved glycan modification of SV2 (green and light blue sticks), and a lipid (brown sticks) — to launch its attack. Source: Rongsheng Jin and Guorui Yao / UCI
A study co-led by Rongsheng Jin, professor of physiology & biophysics at the University of California, Irvine; Min Dong with Boston Children’s Hospital-Harvard Medical School; and Andreas Rummel with the Hannover Medical School in Germany, reveals an important general mechanism by which the pathogen is attracted to, adapts to and takes advantage of glycan modifications in surface receptors to invade motor neurons. Glycans are chains of sugars synthesized by cells for their development, growth, functioning or survival. Results appear June 13 in Nature Structural and Molecular Biology.
“Our findings reveal a new paradigm of the everlasting host-pathogen arms race, where a pathogen develops a smart strategy to achieve highly specific binding to a host receptor while also tolerating genetic changes on the receptor,” Jin said. “And to some extent, this mechanism by which the toxin attacks human is similar to the one that is utilized by some important broad-neutralizing human antibodies to fight viruses, such as dengue viruses and HIV.”




 8613371530291
8613371530291